Puedes ver el enunciado y la respuesta al ejercicio resuelto en el vídeo [si hace clic en este enlace>913].
Indica qué tipo de isomería estructural pueden presentar los pares de compuestos siguientes:
a) pent-1-ino / 3-metilbut-1-ino
b) butanona / butanal
c) butano-2,3-diol / butano-2,2-diol
 EjerciciosFyQ
EjerciciosFyQ
![\chemfig{HC\equiv C-CH(-[6]CH_3)-CH_2-C\equiv N} \chemfig{HC\equiv C-CH(-[6]CH_3)-CH_2-C\equiv N}](local/cache-TeX/bff1638faddb39d50d1907120ffcd416.png)
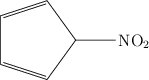
![\chemfig{H_3C-CH(-[6]NO_2)-CH_2-CH=CH_2} \chemfig{H_3C-CH(-[6]NO_2)-CH_2-CH=CH_2}](local/cache-TeX/ab4b29863f03666b22d5852125c4866b.png)
![\chemfig{CH_2=CH-C(=[1]O)(-[7]NH_2)} \chemfig{CH_2=CH-C(=[1]O)(-[7]NH_2)}](local/cache-TeX/3556dcc641ef63a64ef63b1e53f58bbf.png)
![\chemfig{H_3C-CH(-[6]NH_2)-CH_2-C(=[1]O)(-[7]NH_2)} \chemfig{H_3C-CH(-[6]NH_2)-CH_2-C(=[1]O)(-[7]NH_2)}](local/cache-TeX/83fcfab9188e5e4203f117397eae4c3a.png)